What does the word “sugar” really mean? Is it a compound, element, or mixture?
The word “sugar” has different meanings in different contexts. For example, when we talk about sugar as a food additive, it’s an ingredient. When we talk about the sugar content in a fruit juice, it’s a mixture of elements and compounds.

What is sugar?
Sugar is a carbohydrate that metabolizes quickly, giving the body an additional boost of energy to help with tasks like lifting weights. This is because it can act as an instant source of quick fuel for the body. It also helps maintain insulin levels in the blood and therefore helps keep you feeling fuller for longer. When sugar is consumed, it can reduce your appetite and make you less likely to overeat at other mealtimes.
Chemical formula
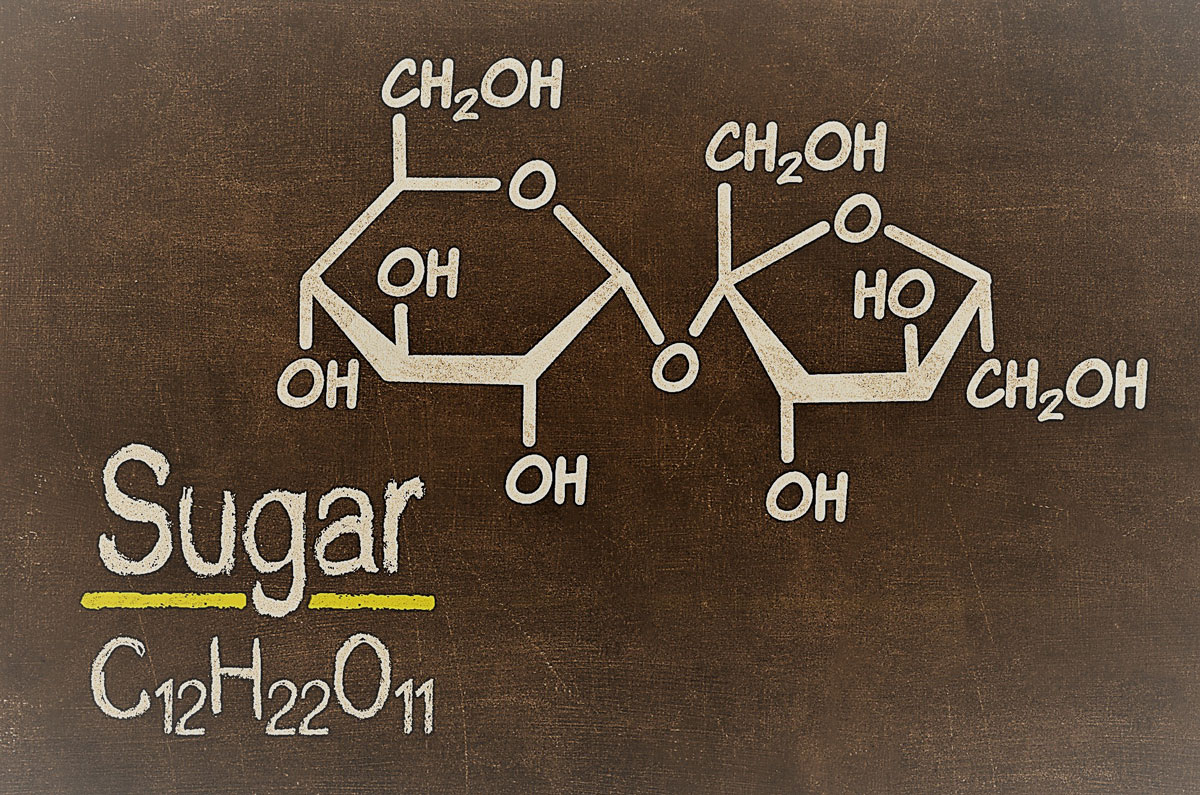
Sugar can be described in its chemical formula, C12H22O11. This means that sugar is made of carbon, hydrogen, and oxygen. The carbons are the center of the sugar molecule which has 6 carbons in total. 6 hydrogens are on each side of the 6 carbons. Oxygen molecules are on the outer end of the 6 carbons with 11 total oxygen molecules per sugar molecule.
A brief history of sugar
The first sugar ever known to man was found in the form of natural cane sugar. It is believed that this sugar originated in India or Asia, where it was made from sugarcane juice. Cane sugar wasn’t discovered until the 1500s by Spanish explorers, but by then other sugars had already been found which are also produced from fruit juice. One form that came about early to rival cane sugar was date sugar, made from pressed dates that were boiled up to extract the juice.
What is a compound?
A compound is a substance composed of two or more elements that cannot be made by themselves.
Compounds can be solid, liquid, gas, or plasma.
The elements in a compound are usually different substances and they form a chemical bond with one another.
What is an element?
Elements are the basic building blocks of matter and consist of one or more protons, neutrons, and electrons. They can be described as atoms that are usually found in the form of molecules.
Atoms are composed of orbiting electrons that are orbiting an atom’s nucleus which consists of protons and neutrons.
What is a mixture?
A mixture is a combination of two or more substances. The substances are mixed to form the mixture. Breakfast cereal is an example of this. It has rice, wheat, corn, whole grain oats, barley, soybean oil, salt, sugar, and artificial colors in it. The cereal itself is made up of many different elements that have been blended to create a single food item.
Is sugar a compound?
Sugar is a compound. Sugar is a compound because it is made up of two different elements, carbon, and oxygen.
Sugar is made up of sugar molecules. One sugar molecule has six carbon atoms, 12 hydrogen atoms, six oxygen atoms, and six nitrogen atoms.
Is sugar an element?
Sugar is not an element but it is classified as a carbohydrate. It is the only carbohydrate that can be absorbed by our body without breaking down. Our body uses sugar to make energy.
Is sugar a mixture?
No, sugar is not a mixture. When thinking about mixtures, there are two types that are possible: heterogeneous or homogeneous. Sugar is an example of a mixture where the components are the same, so it is called a homogeneous mixture.
What are the different forms of sugar?
There are two different forms of sugar: solid and liquid.
- Solid sugar refers to the white sugar crystals that are used for many baking projects. This is the type of sugar used in most recipes for pies, sweet breads, and other desserts.
- Liquid sugar is a form of sugar that is dissolved in water. It is used in baking and cooking to sweeten food, but also can be used in drinks, such as sodas or tea. Liquid sugar gives a softer flavor than rock sugar or table sugar.
FAQ
-
Why is sugar a compound?
It is a compound because it is made up of two or more elements. For example, sugar contains carbon, hydrogen, and oxygen. The carbon atoms are bonded to one another in straight lines called “carbon chains.” The hydrogen atoms are attached to the carbon chains by covalent bonds. Oxygen molecules are also attached to the carbon chains by covalent bonds.
-
Is sugar water a compound?
No, sugar water is not a compound. It is a solution, which means that it can be broken down into its components. When sugar is dissolved in water, the sugar molecules are the solute and are dispersed throughout the water.
-
Is sugar an element or compound?
Sugar is not an element, it is a compound. It’s made up of many different things that are put together. The more sugar you have the more different elements are in it.
-
Is table sugar a compound?
Table sugar is a combination of two molecules: glucose and fructose. These sugars are combined to form the white, powdery substance known as table sugar. It can also be found in its natural state in fruits such as grapes, bananas, and apples. It’s sold in stores in many different forms including powdered and cubed.
-
Is sugar an ionic or covalent compound?
Sugar is a covalent compound. It is not ionic because the bond between the -OH and the COOH groups are not oppositely charged. The sugar molecule is neutral, with each group attracting electrons to create a shared electron cloud.
-
What type of bond is sugar?
A sugar bond is a type of covalent bond that is formed when two atoms of hydrogen and oxygen connect to form a molecule of water, H2O. The bonding pair, the hydrogen and the oxygen, forms a polar covalent bond with opposite charges. The oxygen atom carries a slight negative charge (-) and the hydrogen atom carries a slightly positive charge (+). This means that water molecules will always try to form chains or rings so they can share their electrons.


